The Inflation Reduction Act (IRA), signed into law in August 2022, gave Medicare—for the first time—the authority to negotiate prices for certain high-cost prescription drugs under the Medicare drug price negotiation program. The program began under the Biden Administration and has continued under the Trump Administration, with the aim of lowering out-of-pocket costs and healthcare spending.
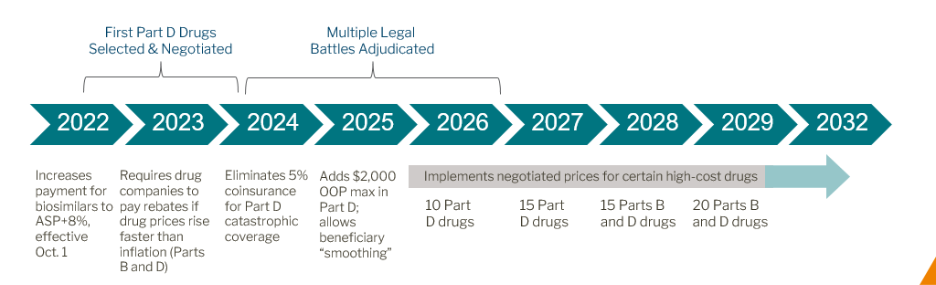
- IPAY 2026: The list of first negotiated prices (Maximum Fair Price, or MFP) for 10 Part D drugs is available here.
- IPAY 2027: The list of 15 additional drugs for negotiation is available here.
- IPAY 2028: Part B drugs will be included for the first time (up to 15 drugs per cycle).
Selection Criteria: Drugs selected for negotiation are based on the following:
- Medicare Spending: Drugs with the highest total spend in Medicare Part B or D.
- Market Exclusivity: Must be single source (no generics or biosimilars).
- Approval Age:
- Small molecules: ≥7 years on market
- Biologics: ≥11 years on market
- Exemptions:
- Orphan drugs with only one indication
- Small biotech drugs (temporary exemption)
- Drugs with low Medicare spend
In the One Big Beautiful Bill Act (OBBA), the ORPHAN Cures (OCA) provision excluded drugs with multiple orphan designations and matching indication(s) from negotiation.
- This effectively resets the clock for when a drug can be subjected to Medicare drug price negotiations. Drugs will become eligible for IRA negotiation 1) 7 years after first NDA approval for NDA drugs or 2) 11 years after first FDA licensure for BLA drugs. If a drug initially qualifies for the orphan exemption but later gains a non-orphan indication, it will not be eligible for negotiation until 7 or 11 years after it no longer qualifies for the orphan exemption, depending on approval type.
Negotiation Process Highlights: The negotiation process takes place in three steps:
- Initial Offer: CMS sends a proposed MFP to the manufacturer based on statutory factors (R&D costs, therapeutic alternatives, market data).
- Counteroffer: Manufacturers may respond with a counteroffer; CMS must reply in writing.
- Final MFP Publication: CMS publishes the negotiated MFP and rationale by March 1 of the year prior to implementation.
Inflationary Penalties
- Excise Tax Penalties: Non-compliance with negotiation terms can trigger excise taxes ranging from 185.71% to 1,900% of the drug’s price.
- The tax applies per unit sold during the period of non-compliance, making it financially prohibitive for manufacturers to opt out.
Out of Pocket Cost Maximum
- Annual Cap for Part D: Starting in 2025, the IRA sets a $2,000 annual cap on out-of-pocket spending for Medicare Part D beneficiaries. This is a major shift from the previous system where costs could be unlimited after catastrophic coverage.
- Monthly Payment Option: Beneficiaries can opt into a “smoothing” program that allows them to spread out their out-of-pocket costs over the year in monthly installments, reducing financial shocks.
- Impact on High-Cost Drugs: This cap significantly benefits patients taking expensive specialty medications, as they will no longer face thousands of dollars in upfront costs early in the year.
- Inflationary Penalty Connection: Manufacturers face penalties for price increases above inflation, which indirectly helps keep beneficiary costs stable.
- Implementation Timeline:
- 2024: $35 monthly insulin cap and $0 cost-sharing for recommended vaccines.
- 2025: $2,000 annual cap fully implemented for Part D.
- 2026 onward: Negotiated drug prices (MFP) further reduce patient costs.
- Beneficiary Enrollment:
CMS will provide guidance on how patients can enroll in the monthly payment program and how plans will operationalize these changes.
Maximum Fair Price
The Maximum Fair Price (MFP) is capped based on a percentage of the drug’s Non-Federal Average Manufacturer Price (non-FAMP), which reflects the average price paid by wholesalers to manufacturers for drugs distributed to nonfederal purchasers, excluding nominal prices.
MFP Ceiling Percentages by Drug Age
| Drug Type | Years Since FDA Approval | MFP Ceiling (% of non-FAMP) |
|---|---|---|
| Small Molecule | 9-12 years | 75% (short monopoly) |
| Any Drug | 12-16 years | 65% (extended monopoly) |
| Any Drug | >16 years | 40% (long monopoly) |
Note: The MFP ceiling percentages listed above are calculated using the drug’s 2021 Non-Federal Average Manufacturer Price (non-FAMP) as the reference point.
Additional Constraints
- For Part B drugs starting in 2028, the MFP cannot exceed the lower of:
- The drugs ASP (Average Sales Price) from the prior year or,
- The applicable non-FAMP percentage ceiling.
- Provider Reimbursement: Set at 106% of MFP, replacing the traditional ASP-based model
Evolution of the Medicare Drug Price Negotiation Program
Since its implementation, policymakers have made a few notable updates to the Medicare drug price negotiation program.
- In the PFS Final Rule for CY 2026, CMS finalized a proposal to stop publishing ASP for these drugs, only listing MPF in the Medicare Part B Drug Payment Limit file.
- The Network remains concerned about the shift from Average Sales Price to MFP, which compresses margins for community practices. Notably, The Network is supportive of H.R. 4299 (Protecting Patient Access to Cancer and Complex Therapies Act) in the 119th Congress, a pending legislative proposal that restore ASP +6% reimbursement for Part B drugs.
